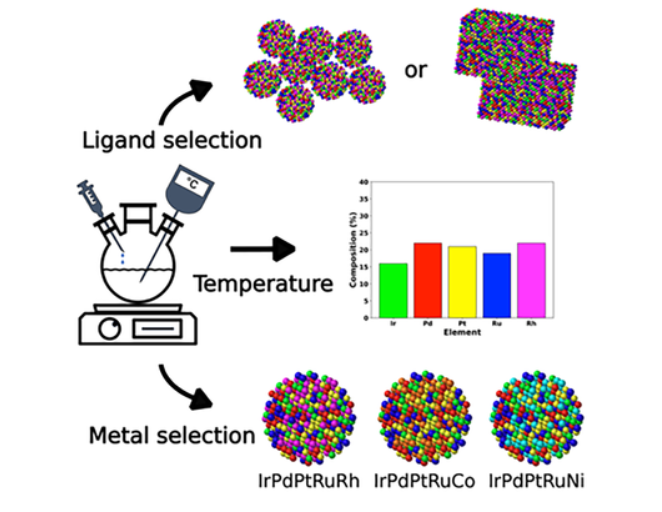
High-entropy alloys (HEAs) continue to gain attention as complex multimetallic systems with promising applications, though their synthesis remains challenging due to the interplay of many parameters. In this study, we explored the synthesis of platinum-group-metal-based HEA nanoparticles using triethylene glycol as both solvent and reducing agent, identifying the hot-injection approach as superior to one-pot synthesis for obtaining phase-pure nanoparticles.
The precursor chemistry strongly influences particle characteristics: metal-chloride precursors yield larger, irregular particles, while metal-acetylacetonate precursors produce smaller, spherical nanoparticles. Pair distribution function (PDF) analysis confirms ultrasmall crystallite sizes of 1–3 nm in both cases, and higher synthesis temperatures promote increased metal incorporation into the alloy structure.
Kinetic studies reveal distinct yet interdependent reduction behaviors among the metals, following the trend Pd > Pt > Rh > Ru > Ir. Substituting Rh with Ni or Fe alters the reduction kinetics of all elements, highlighting their co-dependent nature. Overall, this work provides insight into how key synthesis parameters govern HEA nanoparticle formation, supporting the development of more controlled and tunable colloidal synthesis strategies.
Leave a comment