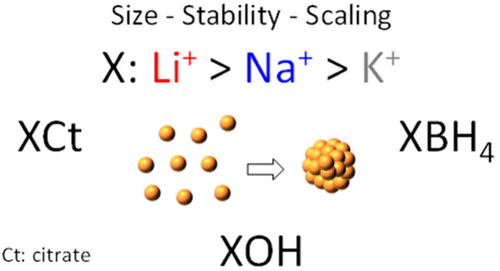
A recent study involving Jonathan Quinson and Espen Drath Bøjesen has uncovered a surprisingly effective way to improve the stability, size, and reproducibility of gold nanoparticles: changing the type of cation in standard synthesis recipes.
The research shows that lithium (Li), compared to commonly used potassium (K) and sodium (Na), produces smaller and more stable nanoparticles—even at higher concentrations. This breakthrough works across different synthesis methods without altering other ingredients, enabling better reproducibility, longer shelf life, and higher-concentration synthesis.
Key findings:
· Li works best across synthesis types
· Improved stability and reproducibility
· Supports higher concentration synthesis
· No changes to other reagents required
The study highlights how ions, often considered background players, can act as powerful design tools in nanoparticle synthesis. It also emphasizes the importance of local chemistry in nanoparticle growth and the need for multi-technique characterization.
Gregory Nordahl and Espen Bøjesen from the Disorder Group contributed advanced STEM and 4D-STEM imaging, which was crucial for validating PDF-based insights into particle shape. Their work demonstrates how combining diverse characterization methods strengthens understanding of nanoparticle formation.
https://pubs.acs.org/doi/10.1021/acs.nanolett.5c04815
Leave a comment